Iron Regulation Part II: Transport Signaling
All these years I was trying to understand the relationship between serum free iron, bound transferrin iron, storage as ferritin iron and anemia. What is the control mechanism? What is the cause of iron deficiency? How is this related to chronic anemia? How much lab data do we need to determine true iron status? OK. So let’s review iron cycling through each body compartment.
Bivalent Iron
You want lab data that measures all these forms of iron transport and storage for a complete and accurate assessment.
Iron exists in two states (bivalent). Ferrous Fe² iron is a reduced state which is more easily absorbed. Ferric Fe³ iron is the more reactive oxidized state. It is less easily absorbed. Ferric iron generates free radicals which can damage tissues and DNA. I will show you later why the more reactive form of iron generates so much controversy.
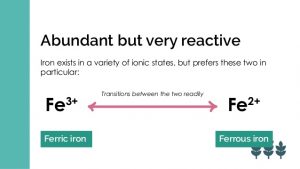
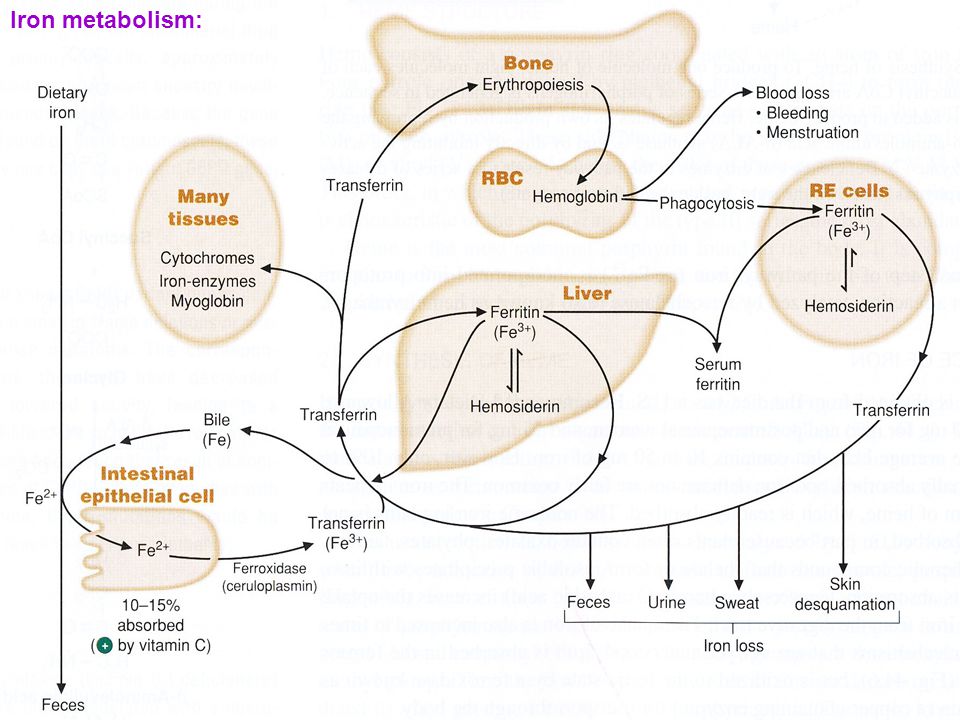
Figure 6 shows the traversal of iron into various key compartments. Here is what is important.
Iron Transport
Dietary iron in the ferrous form is transported across the gut (enterocytes) into the blood stream. Intra-vascular ferrous iron is enzymatically converted to the more reactive ferric iron by ferroxidase. It is then bound to Transferrin. This carrier binding protein transports iron throughout the blood to various tissue sites.
Iron then enters the cells of the liver, spleen, muscle and bone marrow. Inside each cell, iron is bound to Ferritin. This storage protein is capable of binding large quantities of ferric iron. By encapsulating ferric iron, it protects cellular components from reactive free radical damage.
Ferritin is key. I stressed this in part I. Remember, ferritin can also represent inflammation.
The most important compartment is entrance into the bone marrow. That is where red blood cells are manufactured. It is the incorporation of iron into erthryocyte (red blood cell) hemoglobin that is critical for oxygen carrying capacity. Remember, anemia results in oxygen deficit at the cellular level.
Myoglobin is essential for muscle oxygenation. Most especially the muscles of your heart. Hemosiderin is denatured ferritin.
Here is the take home message. You want lab data that measures all these forms of iron transport and storage for a complete and accurate picture.
Hepcidin — Master Controller of Iron Regulation
Now here is my recent discovery and “aha” moment. Hepcidin (1) is the x-factor that I had been missing. It is a liver derived protein that has pro-homonal signaling activity. It is a key regulator of iron absorption. [From the latin hep = liver and cidin = killing.]
Hepcidin, a peptide hormone which is mainly synthesized in the liver, was discovered in 2000. It reduces extracellular iron in the body by several mechanisms: 1) It lowers dietary iron absorption by reducing iron transport across gut mucosal cells (enterocytes); 2) It reduces iron exit from macrophages, the main site of iron storage; and 3) it reduces iron exit from the liver. In all three instances this is accomplished by reducing the transmembrane iron transporter ferroportin. — Wikipedia
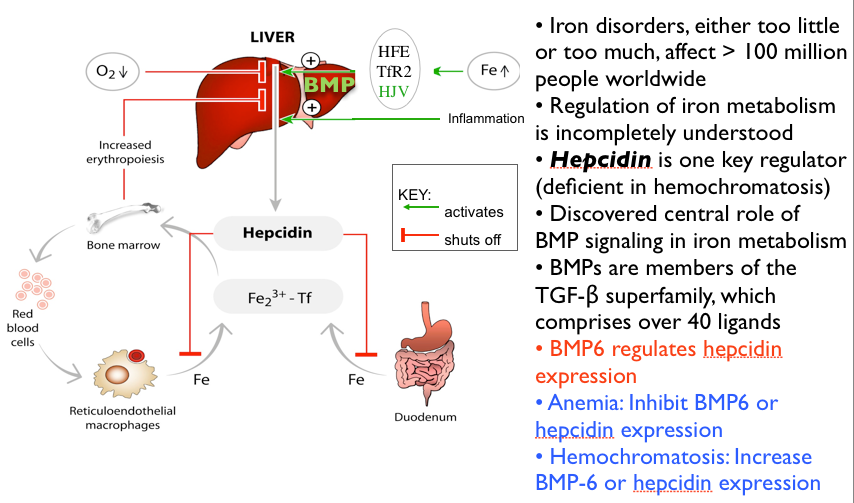
How Hepcidin Controls Iron Absorption
Any signal that increases hepcidin levels decreases iron absorption. Contrariwise, any signal that decreases hepcidin will increase iron absorption
I know. It’s complex picture. But simply put, hepcidin determines efficiency of iron absorption. Any signal that increases hepcidin levels from the liver decrease iron absorption. Contrariwise, any signal that decreases hepcidin will increase iron absorption. You can see in fig 7 that the genetic abnormality hemochromatosis is characterized by a deficiency of hepcidin activity. This causes an accumulation of abnormally toxic iron levels. Inflammation is a major signal for increased hepcidin activity thereby causing anemia.
Hepcidin is a regulator of iron metabolism. Hepcidin inhibits iron transport by binding to the iron export channel ferroportin which is located on the basolateral surface of gut enterocytes and the plasma membrane of reticuloendothelial cells (macrophages)…
Inhibiting ferroportin prevents iron from being exported and the iron is sequestered in the cells.[9][10] By inhibiting ferroportin, hepcidin prevents enterocytes from allowing iron into the hepatic portal system, thereby reducing dietary iron absorption …
Increased hepcidin activity is partially responsible for reduced iron availability seen in anemia of chronic inflammation, such as renal failure.[11] — Wikipedia
That’s a quick overview and background of the complexity of iron transport and control. So how do we correct these deficiencies and improve your iron status? There is a rich array of opinions, guidelines and approaches. That is the subject of the next part III. So stay with me.
- Hepcidin – the Iron Regulatory Hormone, Clin Biochem Rev. 2005 Aug; 26(3): 47–49.